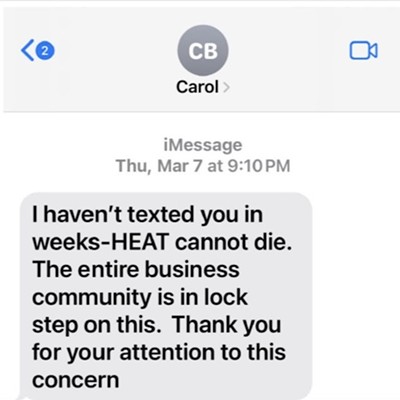
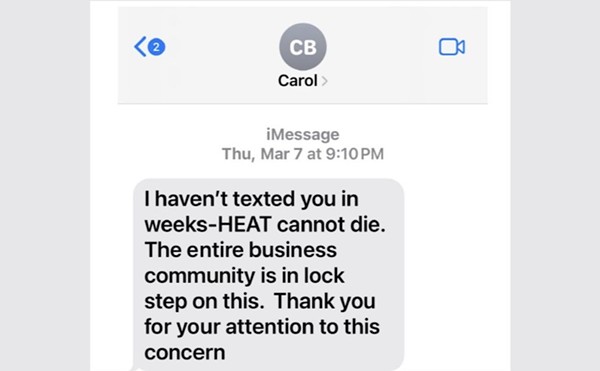
The original ban started in 1983 in the face of the AIDS crisis, back when many still called it the "gay cancer" and little was known about how the human immunodeficiency virus worked. As mostly gay, bisexual and queer men died, the hysteria surrounding the disease reached an all-time high because of a belief that the virus could be spread by casual contact.
"Mothers didn't want me picking up their babies," author Edmund White, a gay man who's had HIV since 1985, told CNN in 2011. "People didn't want to kiss you on the cheek. People certainly didn't want to have sex with you, especially other gay people. It was very isolating and demeaning. That was a long battle."
The FDA ruling indefinitely banned queer men because "of the strong clustering of AIDS illness and the subsequent discovery of high rates of HIV infection in that population," according to the agency. Thousands of recipients of donated blood had already become infected with HIV. As scientists learned more about the virus, they found heterosexual people and children could get HIV, and that it was transmitted through blood, semen and pre-seminal fluids, vaginal fluids, rectal fluids and breast milk. Advanced research also helped develop medication to suppress the virus, prevent transmission and help HIV-positive patients lead healthy lives.
Tests also became much more advanced at screening HIV in donated blood, and it's now a standard practice to test all blood for HIV, hepatitis B, hepatitis C, syphilis and the West Nile Virus. The New York Times reports tests can screen donated blood for the virus "in as little as nine days after the donor has been infected." That "window period" of nine days means donated blood could falsely test negative for HIV if the donor were infected and gave blood within the same nine days, which is the FDA's argument for continuing several bans. The FDA requires blood centers to ask dozens of questions regarding eligibility and defers several populations, including: sex workers, with a permanent ban; injection drug users, with a permanent ban; people who have recently acquired a tattoo or piercing, with a 12-month deferral; people who've completed treatment for syphilis or gonorrhea, with a 12-month deferral; and women who've had sex with queer men, with a 12-month deferral.
The agency says the use of deferral questions, donor education material and HIV donor testing has reduced the risk of HIV transmission through donated blood from one in 2,500 units before standard testing to a current estimated risk of about 1 in 1.47 million transfusions.
"In order to best protect the blood supply in the U.S., the FDA based the December 2015 policy update on what we know about HIV epidemiology in the U.S. today," says FDA spokesperson Tara Goodin in an email. "Previously, there was not enough scientific evidence to support a change in blood donation policy for men who have sex with men (MSM) in the United States. The deferral policy is a behavior-based policy, not one based upon sexual orientation. Current epidemiology shows that a history of male-to-male sexual contact was associated with a 62-fold increased risk for being HIV-positive, whereas the increase in risk for a history of multiple sexual partners of the opposite sex in the last year was 2.3-fold."
LGBTQ advocates argue the 12-month deferral against queer men is discriminatory because straight couples can transmit the virus just as easily, and HIV testing is advanced enough nowadays to easily screen for the virus.